Dear David,
WUFI does not directly output the ice content since this has not been of major interest so far. However, you can compute the ice content from WUFI's standard output.
For liquid water and frozen water to be in equilibrium, they must have the same vapour pressure. Usually, at temperatures below freezing the vapour pressure of ice is lower than the vapour pressure of (subcooled) liquid water because in the ice crystal lattice the water molecules are bound more tightly than in the liquid water. For example, at -10 °C the vapour pressure over (subcooled) liquid water is 286.2 Pa, whereas the vapour pressure over ice is only 259.7 Pa.
However, if the liquid water is exposed to some tension (negative pressure), its vapour pressure is reduced. The capillary forces in the capillaries of a porous material are doing just that: They create a capillary tension in the liquid water and thus reduce its vapour pressure. If at -10 °C the vapour pressure in some capillary is reduced to 259.7 Pa, this specific capillary has just the right radius r0 to create a capillary tension which reduces the vapor pressure of the liquid water to that of the ice at this specific temperature. The equilibrium value of the relative humidity above the meniscus in this capillary and at this temperature is 259.7/286.2 = 0.907 = 90.7 %. In smaller capillaries the tension is higher and thus too high for equilibrium between liquid and ice, and all the water remains liquid at this temperature. In larger capillaries, the tension is too low for equilibrium at the current temperature, and the water is frozen.
In the steady state, the water contained in a porous material collects in the small pores (which have higher 'suction' power) while the large pores remain empty. If at -10 °C all the filled pores have a smaller radius than the r0 described above, all the water contained in the material will remain liquid. If there is enough water in the material to also fill pores with larger radius than r0, the water in those larger pores will be frozen while the water in the pores smaller than r0 remains liquid.
So in order to determine the amount of ice in the material, we must determine the critical pore radius r0 corresponding to the current temperature of the material and see whether the radius of the largest filled pores is smaller or greater than r0. Alternatively, and equivalently, we can express the critical pore size by the capillary tension created in a pore of this size, or by the relative equilibrium humidity above the meniscus in such a pore.
Code: Select all
Temp. p_sat p_sat RH
over over
liquid ice
°C Pa Pa 0...1
0 610.7 610.7 1
-2 527.5 517.3 0.981
-4 454.4 437.1 0.962
-6 390.6 368.4 0.943
-8 334.8 309.7 0.925
-10 286.2 259.7 0.907 < Example
-12 244.1 217.1 0.889
-14 207.5 181.1 0.873
-16 176.0 150.6 0.856
-18 148.8 124.8 0.839
-20 125.4 103.2 0.823
WUFI uses the latter option. Internally it expresses the amount of moisture by the corresponding relative humidity anyway (instead of the water content; the math is easier). To find the amount of ice (if any) at a given location in the material, look at the temperature and the relative humidity at that spot. For -10 °C, the critical relative humidity below which the water will not freeze is 0.907 (= 90.7 %). If the simulation result says that the current relative humidity is less, (say 80 %) all the water is in pores with radii smaller than r0, and it will be liquid.
If the relative humidity is higher (say 0.95 = 95 %), there is water in pores with radii greater than r0, and that water will be frozen. To find the amount of this frozen water, consult the moisture storage function of the material (tabulated in the material data dialog). In Baumberger sandstone, for example, 95 % RH correspond to the moisture content 57.8 kg/m³. This is the _total_ water content. Liquid water is found in the pores with radius less than r0, and according to the moisture storage function the RH of 90.7 % corresponds to the water content 43.9 kg/m³. The difference 57.8 - 43.9 = 13.9 kg/m³ is frozen water.
Please keep in mind, however, that while the approach described above is taken from standard literature (and apparently in agreement with measurements on soils; Neiss J. 1982), it may be a bit simplistic. For example, it assumes that in contrast to the liquid water which is exposed to the capillary tension, the ice remains at atmospheric pressure, and I am not sure to which degree this is really justified for porous building materials as opposed to porous soils. Also, WUFI treats the freezing as a simple phase change; possible pumping effects (as in frost heaves) and similar effects which may affect the amount of ice are neglected. WUFI takes freezing into account as a "secondary" effect only which somewhat affects the simulation result by releasing some latent heat and by reducing the amount of moveable water. WUFI is not meant to explicitly and exactly simulate the freezing process and the resulting amount and distribution of ice.
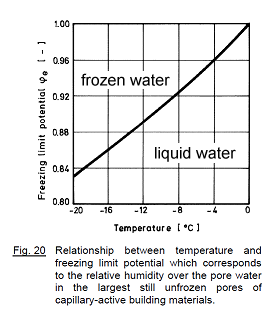
The following diagram shows the critical humidity which separates frozen from liquid water:
- Forum _ freezing limit potential _ small + label.png (34.13 KiB) Viewed 6378 times
This curve is almost a straight line, and WUFI computes it using the simplified fomula
RH_limit = 1 + (temperature, °C)/100. For example, for a temperature of -10 °C this formula gives RH_limit = 0.90, to be compared with the value 0.907 shown in the table above.
Regards,
Thomas